Abimbola Amoo1,2,* and Hannah O Dada-adegboola1
- Department of Medical Microbiology and Parasitology, University College Hospital, Ibadan, Oyo State, Nigeria
- Department of Epidemiology and Population Health, University of Louisville, Kentucky, USA
Received Date : 16 March 2023 , Accepted Date : 20 April 2023 , Published Date : 8 May 2023
*Correspondence Address:
Abimbola Amoo, Department of Medical Microbiology and Parasitology, University College Hospital, Ibadan, Oyo State, Nigeria; Department of Epidemiology and Population Health, University of Louisville, Kentucky, USA.
Copyright©2023 by Amoo A, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Background: Malaria could lead to severe deaths, if the COVID-19 pandemic disrupts core strategies, according to a recently released WHO modeling analysis report. Transfusion-transmitted infections (TTIs) are mainly studies of the prevalence of bacteria and viruses with very little or no effort to investigate the parasitic malaria infection that can cause disease after blood transfusion, especially in patients with hereditary hemolytic anemia. This study was designed to investigate the effects of storage on Plasmodium parasitemia.
Case presentation: A Cross-sectional study in Voluntary blood donors. Demographic data and clinical history were obtained using a pro-formal questionnaire. Blood samples were collected from blood donors and then stored at 4°C for 21 days. Each sample was examined for P. falciparum trophozoites microscopy on the initial day, that is day 0 and the three subsequent follow-ups at day 3, 7, and 21 using giemsa stained light microscopy.
Result: A total of 248 blood samples collected were collected among study participants and tested for Plasmodium falciparum. The overall prevalence of Plasmodium falciparum infection using the Giemsa microscope was 8.5%. The prevalence of blood samples stored for 3, 7, and 21days was 8.1%, 7.3%, and 5.7%, respectively. Microscopy showed a significant reduction in the prevalence and density of Plasmodium falciparum with an increased blood storage duration (Kendall’s tau; p<0.001).
Conclusions: This study shows that the prevalence of Plasmodium falciparum decreases with increasing storage days. The impact of storage on the longevity of red blood cells in donated blood needs to be evaluated.
Keywords:
Malaria; Transmissible; Surveillance; Storage effects.
Introduction
Human malaria is a mosquito-borne infectious disease caused by intraerythrocytic protozoa of the genus Plasmodium namely; P. falciparum, P. vivax, P. ovale, P. malariae and P. knowlesi [1].Plasmodium species indeed require two hosts to complete their life cycle, with humans acting as the intermediate host and mosquitoes acting as the definitive host [2]. Among the different species of malaria, P. falciparum is the most widespread and is responsible for most malariarelated deaths worldwide [3]. Moreover, P. falciparum infections can often be resistant to chloroquine treatment, which is a commonly used antimalarial drug. The mosquito Anopheles has a sexual cycle and human beings have a two-phase cycle involving red blood cells (erythrocytic phase) and the cell that makes up the liver, the hepatocytes (pre-erythrocytic phase) [4]. In the case of mosquito-transmitted malaria, the incubation period is about 1-2 weeks but for the case of transfusion-transmitted malaria, the period is about 2-4 days [5]. This is partly because blood parasites transmitted through
blood transfusion bypass the extra red blood cell cycle extra erythrocytic cycle. In endemic areas including Nigeria, the risk of contracting malaria transmitted by blood transfusion is about 1-50 cases per million donated units [6]. The factors influencing the prevalence of blood transfusion-transmitted malaria include seasonal variations with increase in incidence observed during the rainy season, blood donor selection criteria, and blood donor screening methods [7]. In Nigeria, Plasmodium falciparum is the most common cause of malaria infections transmitted through blood transfusion [8].
Many blood donors in endemic areas are asymptomatic carriers of malaria, and their parasite density is often below the detection threshold of currently available diagnostic methods [9]. Individuals with a partially protective immune state are often the source of Transfusion Transmissible Malaria, and the parasites found in the thick blood film are equivalent to approximately 10,000 parasites in
450ml blood units [10]. Inoculations of Plasmodium species from transfused blood can pose a greater threat than bites from infected mosquitoes, with only 15 parasites required to cause a blood transfusiontransmitted malaria infection. Plasmodium species can survive in blood cells for about 21 days outside the host [11]. In Nigeria, screening for malaria parasites is not regulated by current national blood transfusion guidelines [12]. This is because transfusion-induced malaria transmission is not generally considered a serious problem in adults whose immune levels are considered to be effective in combating transfusion-induced malaria in endemic areas [13]. However, many health conditions that require blood transfusions, such as road accidents, pregnancy-related hemorrhage, and severe immunosuppression, increase the chances of the transmission of blood infection [13,14]. Malaria parasite sensitivity decreases with low parasite densities found in the blood of infected asymptomatic carriers. It is not sensitive enough for blood donor screening [15]. Other methods of diagnosis include immunoassays such as indirect hemagglutination, immunofluorescent assay, radioimmunoassay, enzyme-based latex immunosorbent assay, and antigen-based rapid diagnostic test, as well as molecular amplification methods such as using agar-gel diffusion and immunoassay to detect P. falciparum antigen and DNA [14].
Methods
Nature of the study
The study was a cross-sectional study of 248 non- remunerated voluntary blood donors at the National Blood Transfusion Service Center in Ibadan, Oyo, Nigeria. This study was conducted from June 2019 to February 2020.
Using a random sampling technique, 248 consented participants between the ages of 18 and 59 were selected and randomly recruited following their written informed consent. The study participant who had been treated for malaria in the past month and had less than 30% packed red blood cells (not suitable for donors) was excluded from the study. Participant whole blood samples were stored at 4°C, and then taken from the blood bag cord, the samples were dispensed into EDTA bottles. Samples were giemsa stained and each participant’s slides were examined for Plasmodium falciparum under a light microscope. The examination of Plasmodium falciparum tests was performed 0,3,7, and 21 days after sample collection.
Laboratory investigation for P. falciparum by Microscopy
Thick blood smears were prepared from each fresh donor blood sample, air dried and then stained with freshly prepared 10% giemsa stain for 10 minutes. The stains were gently washed, the slides were air dried, and then inspected with an X100 oil immersion microscope magnification.
All positive samples on the initial day 0 were repeatedly tested on day 3 (f0), day 7 (f7), and day 21 (f21). For positive slides, the number of parasites was counted against 200 leukocytes, and quantification of parasite density estimated by assuming 8,000 leucocytes/µl of blood, after been examined by two microscopist and average score was taken. Slides were reported negative if no Plasmodium falciparum was detected in 100 fields of each thick smear. A sample known to have a high number of Plasmodium falciparum trophozoite was used as a positive control.
Data collection
The data were collected using a self- administered, semi-structured questionnaire. Section A consists of data on the collected socio- demographic characteristics, and Section B are information such as mosquito net use, living river areas, blood types, and malaria prophylaxis use.
Data management and statistical analysis
Collected data were coded, cleaned and computed into Microsoft Excel spreadsheet and double checked for accuracy. Data on prevalence of P. falciparum were analyzed using STATA version (Stata Corporation, College Station, Texas, USA). The categorical variables were summarized as proportions and frequencies. Test of association between/among qualitative variables was done using bivariate and multivariate analyses. The effect of storage duration on P. falciparum density was compared using Kendall’s Tau test, with the level of statistical significance set at a p-value <0.05.
Results
A total of 248 questionnaires were administered to non-remunerated blood donors. The blood film microscopy of the respondents was analyzed for Plasmodium falciparum trophozoite positivity at the on day 0 was Twenty-one (8.5%). Two hundred (80.6%) out of the respondents were male, and one hundred and six (42.7%) were between the ages of 29 and 39. One hundred and twenty-two (49.1%) respondents reported sleeping under mosquito bed nets, and only 4 (1.6%) had taken malaria prophylaxis. As shown in Figure 1 and Table 1.
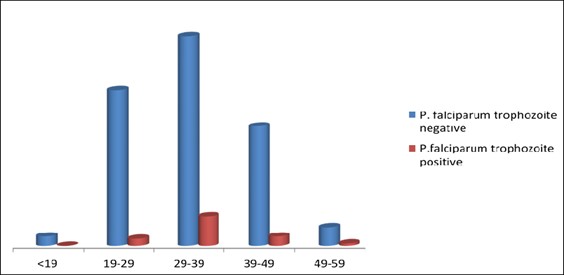
Figure 1: P. falciparum positivity and negativity distribution with age.
|
Risk Factor |
MP Negative n(%) |
MP Positive n(%) |
X2 test |
P value |
| 1. Malaria prophylaxis |
|
|
0.376 |
0.54 |
| No |
223(98.2%) |
21(100%) |
|
|
| Yes |
4(1.76%) |
0(0%) |
|
|
| 2. Mosquito bed nets |
|
|
1.131 |
0.202 |
| No |
113(49.8%) |
13(61.9%) |
|
|
| Yes |
114(50.22%) |
8(38.1%) |
|
|
| 3. Level of Education |
|
|
0.314 |
0.78 |
| High School |
20(8.81%) |
2(9.52%) |
|
|
| Undergraduate |
74(32.6%) |
8(38.1%) |
|
|
| Graduate |
133(58.6%) |
11(52.38%) |
|
|
| 4. Blood groups |
|
|
3.751 |
0.189 |
| A |
26(11.45%) |
4(19.1%) |
|
|
| AB |
5(2.20%) |
1(4.76%) |
|
|
| B |
25(11.0%) |
0(0.00%) |
|
|
| O |
171(75.33) |
16(76.2%) |
|
|
Table 1: Risk factors associated with P. falciparum infection.
Risk factors associated with Plasmodium falciparum infection
Among the 21(8.5%) participants who were positive for P. falciparum trophozoites, 8(38.1%) slept under insecticide treated bed nets (ITNs) and this was found to be not statistically significant (p=0.202). Furthermore, no significant association was observed among those that had antimalaria prophylactics (p=0.540), level of education (p=0.780), and blood groups (p=0.189).
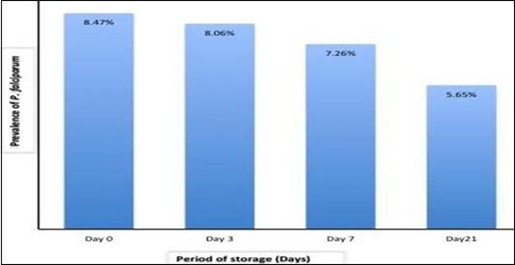
Figure 2: Prevalence of P. falciparum infection in follow-up stored blood samples by Microscopy.
There was a significant difference in the prevalence of Plasmodium falciparum infection in the results of the microscopic examination (8.5%) of the study participants on initial day 0. There was a significant difference in the prevalence of Plasmodium falciparum infection (8.1%) on the day 3 (p <0.001). The infection rate of Plasmodium falciparum on the day 7 was 7.3% (p<0.001). The infection rate of P. falciparum was 5.7% on day 21 (p<0.001) (Figure 2).
Effect of storage duration on P. falciparum density
During the baseline examination on day 0 and three subsequent observations on days 3,7 and 21, the parasite density for the initially positive microscopic specimens ranged from 75 to 8679 parasites/μL of blood. At the first observation, the density of parasites ranged from 37 to 7885 parasites/μL of blood, the 3-day storage period was associated with a significantly lower density of parasites (Kendall’s τb=0.9738, p <0.001). At the second observation after 7 days, the density of parasites ranged from 36 to 3680 parasites/μl of blood, the storage period was associated with a significantly lower density of P. falciparum (Kendall’s τb=0.9196, p <0.001). in the third observation (14/248) there were 15 to 379 parasites/μL of blood. A shelf life of 21 days was associated with a significantly lower density of the P. falciparum parasite (Kendall’s τb=0.8042, p <0.001). Increase in storage period was associated with a significantly lower density of P. falciparum.
Discussion
Blood from each blood donors was taken and examined for malaria parasites before being stored at 4°C. Some of the blood samples were stored at 4°C and examined for P. falciparum trophozoites after 3,7, and 21 days using microscopy. The results of the study showed that the prevalence of P. falciparum was lower on Day 0 among blood donors in Ibadan, Nigeria, when compared with similar study in Nigeria [16]. In Ghana, Owusu-Ofori, et al. reported 13.7% of patient blood donors. The results of the microscope examination of the study participants showed that the prevalence of P. falciparum was low [17]. The study was carried out before the rainy season from January to April when malaria transmission is low [18].
Cold storage at 4°C has a significant effect on the prevalence and density of Plasmodium falciparum in blood samples. Specifically, it indicates that the prevalence of P. falciparum decreased slightly on the 3rd day of storage compared to the initial day and that this trend continued on the 7th and 21st days, with a steady decrease in parasite density. These results are consistent with previous research, which has shown that cold storage can have a significant impact on the viability and infectivity of malaria parasites in blood samples. Cold temperatures can slow the metabolic activity of the parasite and reduce its ability to replicate and cause disease [19].
However, it is important to note that the extent and duration of the effects of cold storage on P. falciparum can vary depending on several factors, including the initial parasite load, the storage conditions, and the duration of storage.20 Therefore, it is critical to carefully monitor and manage the storage of blood samples to ensure that they remain viable and safe for use in diagnostic and therapeutic applications. Prevalence of P. falciparum decreased slightly on the 3rd day of storage at 4°C compared to initial day 0, and the prevalence of P. falciparum on the 7th and 21st days. It steadily decreased to 7.2% and 5.4%, respectively. Parasite density decreases with increasing storage period and indicating that cold storage has a significant effect on P. falciparum [20].
There is no evidence to suggest that most blood donors are graduates, and this demographic group is not necessarily associated with a reduced risk of P. falciparum. While education may play a role in blood donation, it is not a determining factor and many blood donation campaigns and programs aim to engage a diverse range of donors from different backgrounds and educational levels. It is important to focus on effective screening and testing procedures to prevent malaria transmission through blood transfusions, regardless of the educational background of blood donors [21].
Conclusion
The overall prevalence of P. falciparum in the blood sample on day 0, first follow-up after 3 days, second follow-up after 7 days and third follow-up after 21 days of storage were 8.4%, 8.1%, 7.5%, and 5.4% respectively. This implies that longer periods of storage were associated with significant lower densities and prevalence. The risk of P. falciparum transmission can be reduced by keeping blood for longer.
Acknowledgment
The authors are indebted to our mentor Professor R.A. Bakare of the Department of Medical Microbiology and Parasitology, and Professor Olaniyi John and the Staff of Hematology and Blood Transfusion Department, University College Hospital, Ibadan, Nigeria for their support in the collection of data.
Ethical considerations
Ethical approval to conduct this study was obtained from the Oyo State Ethical Review Committee and UI/UCH Ethical Review Committee in Oyo State, Ibadan, Nigeria. This research was conducted in strict compliance with the guidelines of the 1964 Declaration of Helsinki. Written and verbal informed consent was obtained from all study participants before they are enrolled in the study.
Conflict of interest
All the authors declare no conflict of interest.
References
- World Health Malaria. Fact Sheet. 2023.
- Rogerson SJ, Desai M, Mayor A, Sicuri E, Taylor SM, van Eijk AM. Burden, pathology, and costs of malaria in pregnancy: new developments for an old problem. Lancet Infect Dis. 2018;18(4):e107-18.
- Nosten F, White NJ. Artemisinin-based combination treatment of falciparum malaria. Defining and Defeating the Intolerable Burden of Malaria III: Progress and Perspectives. Am J Trop Med Hyg. 2007;77(6):181-92.
- Centers for Disease Control and Prevention. Malaria-Transmission.
- Golassa L, Erko B, Baliraine FN, Aseffa A, Swedberg G. Polymorphisms in chloroquine resistance-associated genes in Plasmodium vivax in Ethiopia. Malar J. 2015;14(1):1-5.
- World Health World Malaria Report. 2017.
- Mangano VD, Perandin F, Tiberti N, Guerriero M, Migliaccio F, Prato M, et Risk of transfusion-transmitted malaria: evaluation of commercial ELISA kits for the detection of anti-Plasmodium antibodies in candidate blood donors. Malaria
- 2019;18(1):1-9.
- Mac PA, Asheadzi HF, Gideon A, Thaker P, Airiohuodion Prevalence of Plasmodium falciparum among Nigerians in Abuja and Central States: A comparative analysis of sensitivity and specificity using rapid diagnostic test and microscopy as tools in management of malaria. Int J Trop Dis. 2019;1:14.
- Abdullah S, Karunamoorthi Malaria and blood transfusion: major issues of blood safety in malaria-endemic countries and strategies for mitigating the risk of Plasmodium parasites. Parasitol Res. 2016;115:35-47.
- Hemingway J, Shretta R, Wells TNC, Bell D, Djimdé AA, Achee N, et al. Tools and Strategies for Malaria Control and Elimination: What Do We Need to Achieve a Grand Convergence in Malaria? PLoS Biol. 2016;14(3):e1002380.
- Sato, S. Plasmodium-a brief introduction to the parasites causing human malaria and their basic biology. J Physiol Anthropol. 2021;40(1):1-3.
- Aneke JC, Okocha CE. Blood transfusion safety; current status and challenges in Nigeria. Asian J Transfus Sci. 2017;11(1):1.
- Ekwunife CA, Ozumba NA, Eneanya CI, Nwaorgu OC. Malaria infection among blood donors in Onitsha urban, Southeast Nigeria. Sierra Leone J Biomed Res. 2011;3(1):21-6.
- Eze, UA, and Eze Emerging Molecular Methods for the Diagnosis and Epidemiological Study of Parasitic Infections. ASCII J Health. 2015;2(4):32-43.
- Slater HC, Ding XC, Knudson S, Bridges DJ, Moonga H, Saad NJ, et Performance and utility of more highly sensitive malaria rapid diagnostic tests. BMC Infect Dis. 2022;22(1):1-3.
- Olawumi HO, Fadeyi A, Babatunde SK, Akanbi II AA, Babatunde AS, Sani MA, et al. Malaria parasitaemia among blood donors in Ilorin, Nigeria. Afr J Infect Dis. 2015;9(1):10-3.
- Owusu-Ofori, Alex K, Parry J, Martha B, Christopher M. et al. Transfusion- transmitted malaria in Ghana. Clin Infect Dis. 2013;56(12):1735-41.
- Chaponda EB, Chandramohan D, Michelo C, Mharakurwa S, Chipeta J, Chico High burden of malaria infection in pregnant women in a rural district of Zambia: a cross-sectional study. Malaria J. 2015;14(1):1-2.
- Chattopadhyay R, Majam VF, Kumar Survival of Plasmodium falciparum in human blood during refrigeration. Transfusion. 2011;51(3):630-5.
- Kitchen AD, Chiodini Malaria and blood transfusion. Vox Sang. 2006;90(2):77-84.
- Liu L, Oza S, Hogan D, Chu Y, Perin J, Zhu J, et Global, regional, and national causes of under-5 mortality in 2000- 15: an updated systematic analysis with implications for the Sustainable Development Goals. Lancet. 2016;388(10063):3027-35.